Quality and Environment
Environmental strategy
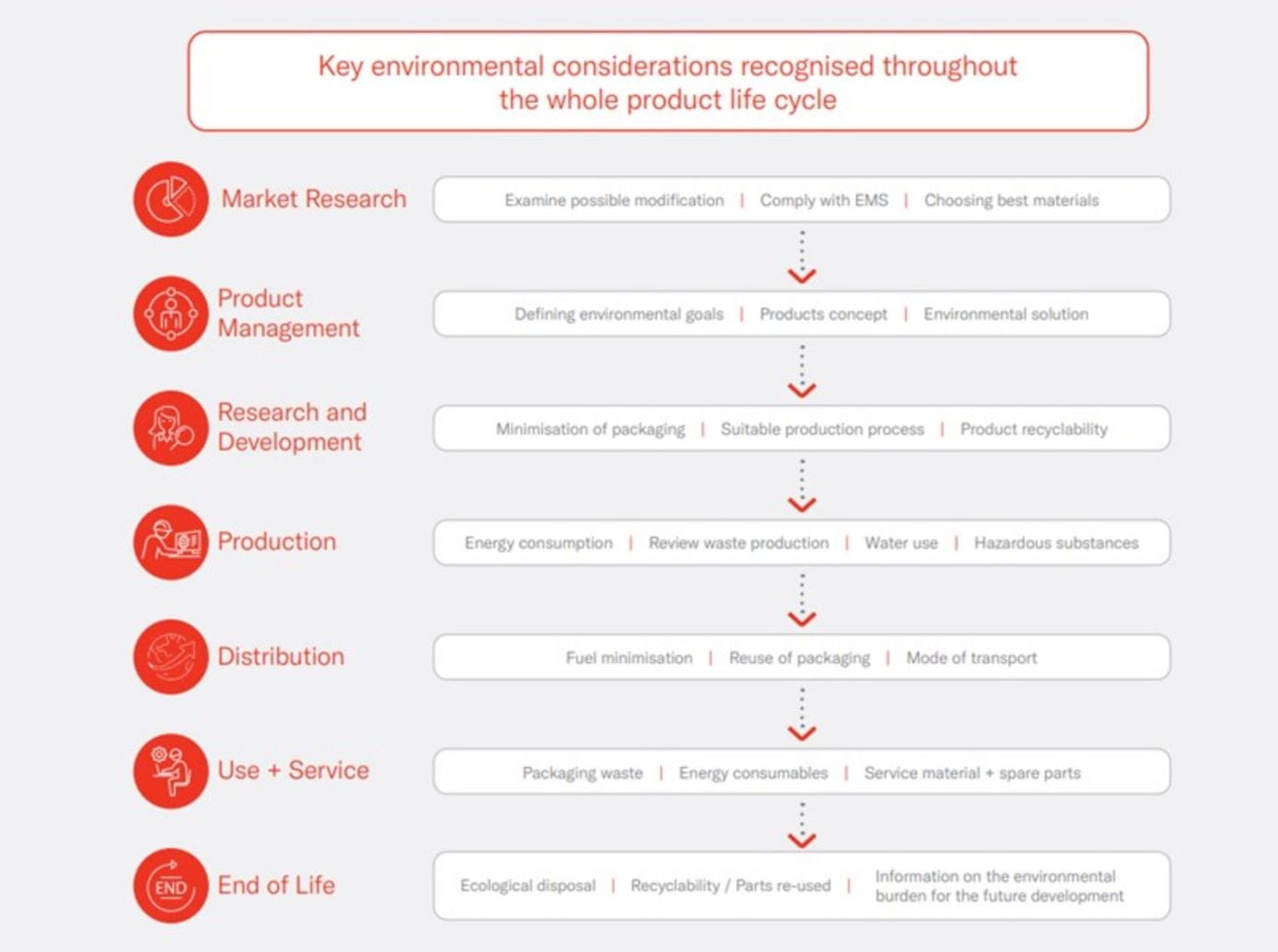
Thinking responsibly throughout the whole product life-cycle process.


Initiatives

Waste management
Only 11% of LINET’s waste production goes to landfill, 85% is recyclable waste, and 4% of waste is used for energy.

Water

Materials and Packaging

Transport and Shipping
We are working on creating a distribution network that is efficient, safe and environmentally friendly – we are committed to reducing fuel consumption, so we are working with suppliers on two-sided improvements as much as possible.
We also encourage using low-carbon corporate cars and actively encourage employees to ride bicycles or cars together and use public transport.
Carbon footprint

Compliance with law and regulations
Certificate ISO 14001:2015 LINET
PDF Document
Certificate ISO 13485:2016 LINET
PDF Document
Certificate ISO 14001:2015 LINET
PDF Document
Cerificate ISO 9001:2015 LINET
PDF Document
Certificate ISO 13485:2016 BORCAD
PDF Document
Certificate ISO 14001:2015 BORCAD
PDF Document
Certificate ISO 9001:2015 BORCAD
PDF Document
MDSAP LINET
PDF Document
IMS Company Policy
PDF Document

